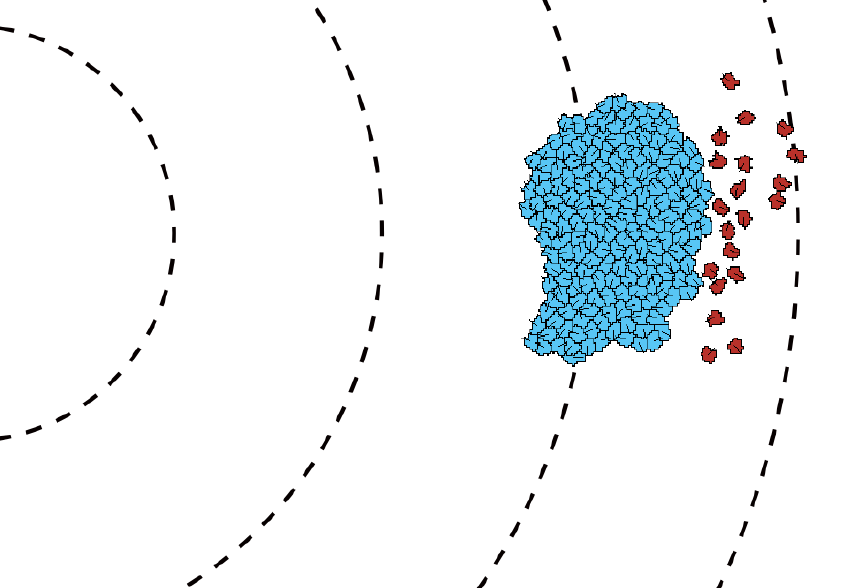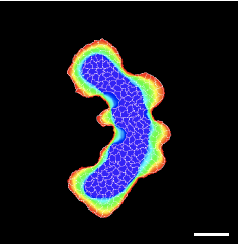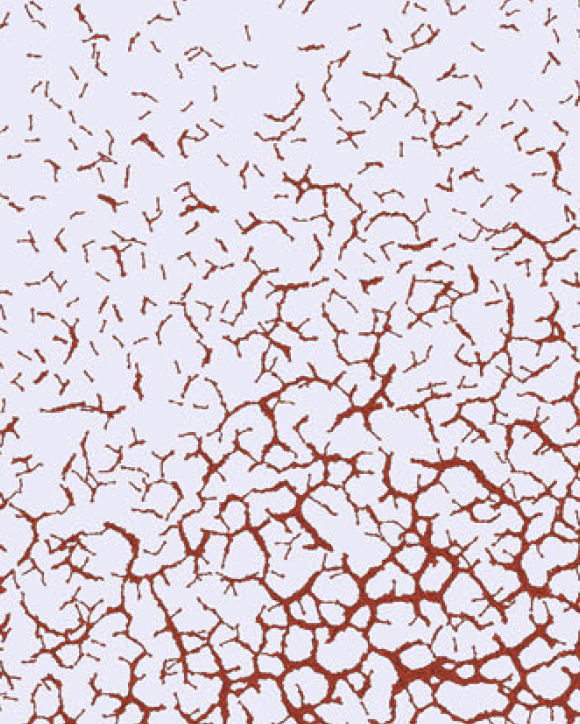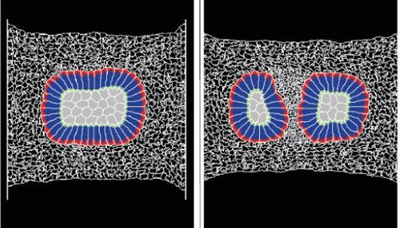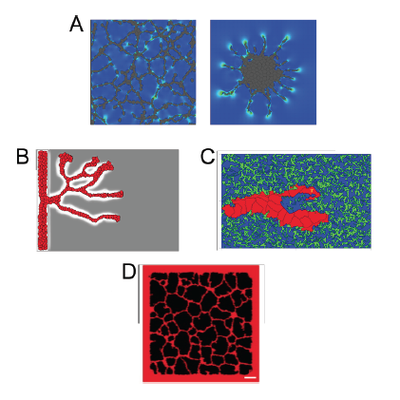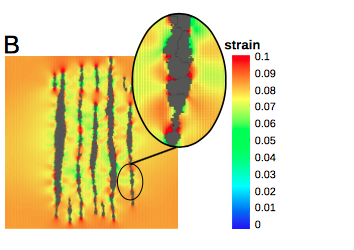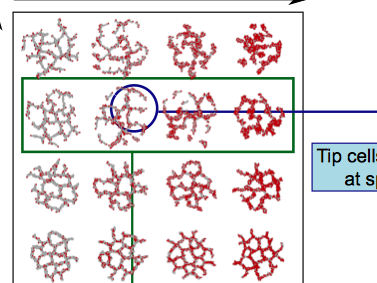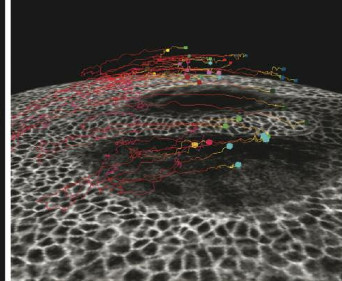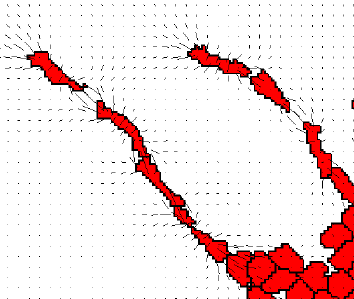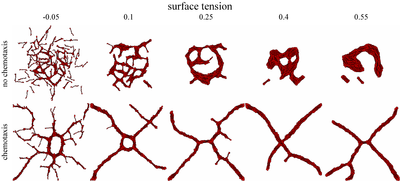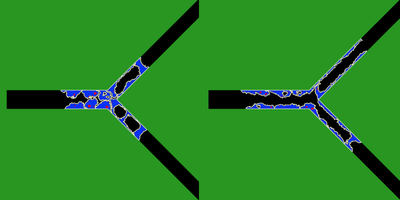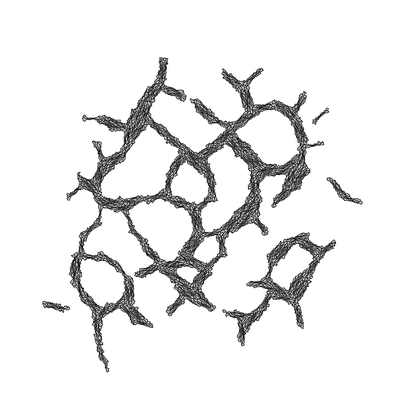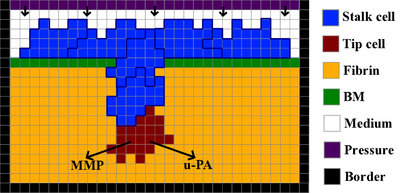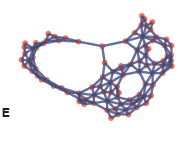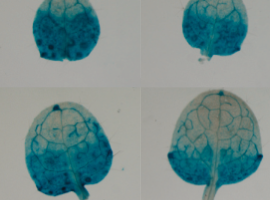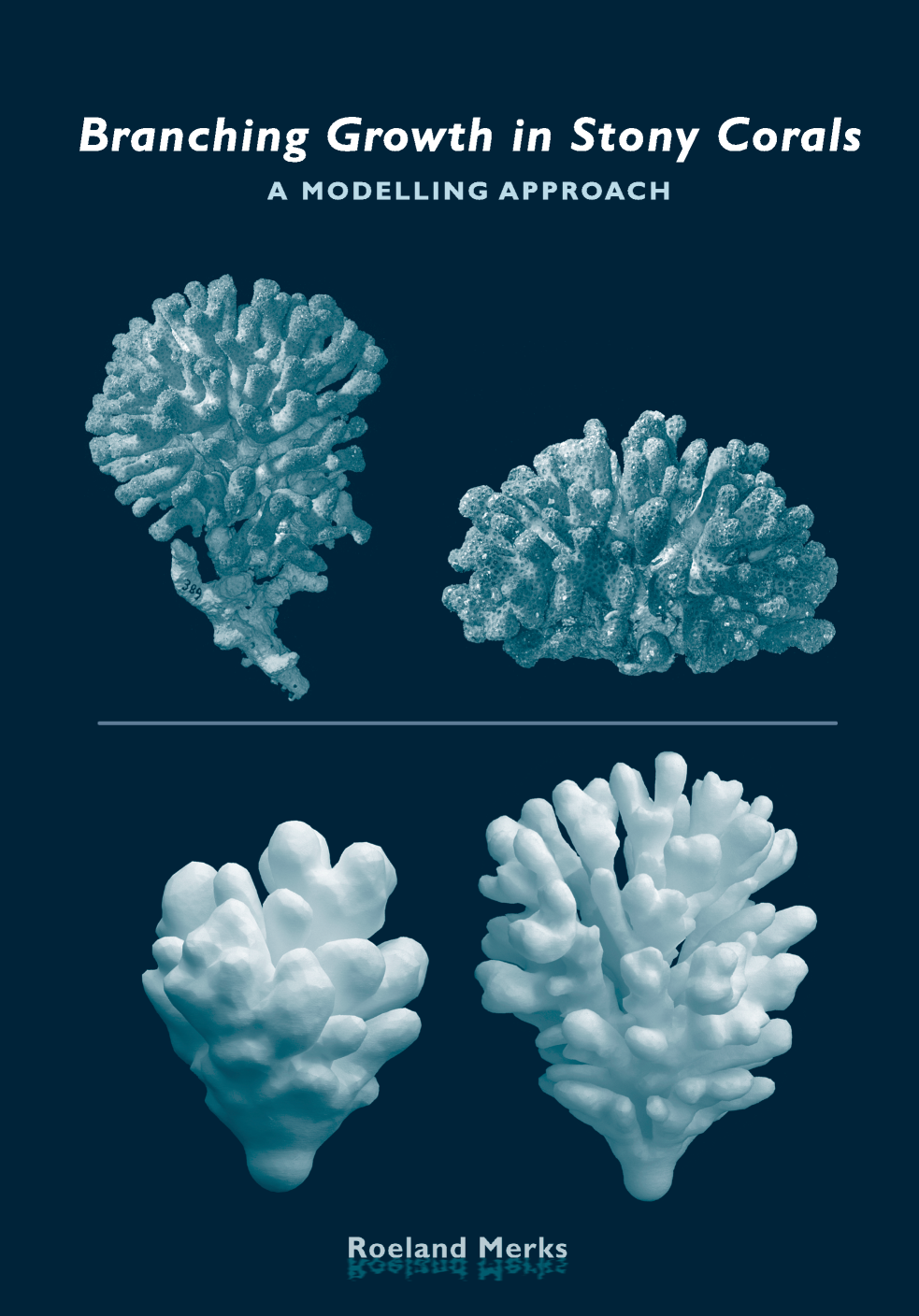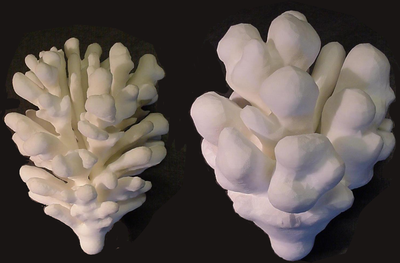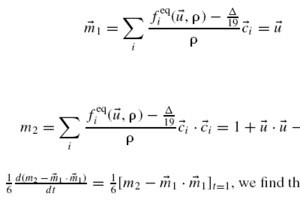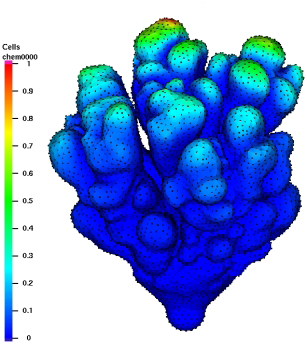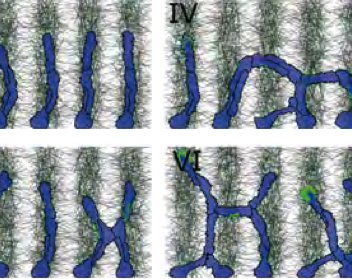
TTo form new sprouts during angiogenesis, endothelial cells coordinate migration through biophysical and biomechanical signaling with each other and the micro-environment. In zebrafish embryos, a key example of this process is intersegmental vessel (ISV) formation, where endothelial tip cells sprout dorsally from the dorsal aorta, elongate between somites, and connect to form the dorsal longitudinal anastomotic vessel (DLAV). While various factors coordinate ISV pathfinding, such as vascular endothelial growth factor (VEGF), semaphorin signaling, and integrin-mediated adhesion, the role of extracellular matrix (ECM) mechanics and distribution remains incompletely understood. Here, we combined in vivo timelapse imaging with mathematical modeling to study how ECM components influence endothelial migration. Experimentally, morpholino single knockdowns of laminin or fibronectin slowed ISV elongation but most vessels eventually reached the DLAV. To analyze potential effects of ECM mechanics on sprout progression, we developed a hybrid mathematical model of ISV elongation. The model combines a mass–spring based representation of a network of ECM fibers with an experimentally validated Cellular Potts Model of endothelial cell behavior that described cell elongation and migration, integrin-based mechanosensitive adhesion formation, and chemical signaling through VEGFs and semaphorins. For baseline parameters, this model predicts that the ECM helps to confine the extending sprout to the intersegmental space. After reducing the stiffness of the ECM network, ISVs sprouted more slowly than in baseline conditions, consistent with our experimental observation. After further reduction of ECM stiffness, the model predicted increased fusion of ISVs, a behavior reminiscent of the behavior of endothelial cells in many in vitro models and in our previous in silico models. Guided by these data and predictions, we hypothesized that ECM in the intersomitic space guides endothelial selforganization during ISV pathfinding. In agreement with this hypothesis and model predictions, combined knockdown of laminin and fibronectin produced network-like endothelial arrangements and errant pathfinding of ISVs. Importantly, re-expression of fibronectin using chimeric fibronectin mRNA substantially restored ISV organization in the triple knockdown backgrounds, supporting that the severe disorganization reflects specific loss of fibronectin-dependent function rather than nonspecific injection effects. Altogether, our results suggest a model of guided self-organization for ISV formation, in which the ECM and chemical guidance cues including semaphorins confine self-organized endothelial network formation to the intersegmental space.